Patient-specific radiomic feature selection with reconstructed healthy persona of knee MR images
New method combines interpretable "radiomic features" with AI-generated "healthy scans" for better, explainable medical image analysis.
Background & Academic Lineage
The problem of diagnosing knee injuries, such as Anterior Cruciate Ligament (ACL) or meniscus tears, has historically relied on physical examinations with limited accuracy. Magnetic Resonance (MR) imaging serves as the clinical gold standard, but automated analysis using end-to-end Deep Learning (DL) models often functions as a "black box." This lack of transparency prevents clinicians from understanding why a model makes a specific diagnosis, which is a critical pain point in medical decision-making. While classical radiomics (hand-crafted features like intensity or shape) offer high interpretability, they often underperform compared to complex DL models. The authors sought to bridge this gap by creating a framework that maintains the interpretability of radiomics while achieving performance comparable to state-of-the-art DL.
Intuitive Domain Terms
- Radiomics: Think of this as "digital fingerprinting" for medical images. Instead of just looking at a picture, the computer extracts hundreds of tiny, specific mathematical measurements (like texture, shape, or intensity patterns) that are too subtle for the human eye to quantify consistently.
- Healthy Persona: Imagine a doctor trying to diagnose a broken bone by comparing the injured limb to a "perfect" version of that same patient's limb. The authors use AI to "imagine" what the patient's knee would look like if it were perfectly healthy, providing a baseline to highlight exactly where the pathology deviates from the norm.
- Denoising Diffusion Probabilistic Model (DDPM): Think of this as a highly sophisticated "image restorer." It learns the statistical patterns of healthy knees by practicing how to remove "noise" (random static) from images until only a clear, healthy structure remains. It uses this skill to reconstruct the "healthy persona" from a damaged input.
Notation Table
| Notation | Description |
|---|---|
| $\mathbf{x}$ | The input image patch containing the region of interest (ROI). |
| $\mathbf{f}$ | The pool of $F$ radiomic features extracted from the image. |
| $g_\theta(\mathbf{x})$ | The feature-weighting neural network with parameters $\theta$. |
| $\mathbf{p}$ | The predicted probabilities for selecting specific features. |
| $\mathbf{f}^w$ | The weighted feature vector used for classification, $\mathbf{f}^w = \{p_i f_i\}_{i=1}^F$. |
| $r_\phi(\mathbf{f}^w)$ | The logistic regression classifier with parameters $\phi$. |
| $\mathbf{x}^{\text{persona}}$ | The synthesized healthy version of the patient's image. |
The authors solve the problem of feature selection by treating it as an optimization task where the model learns to assign importance weights to features. The goal is to maximize the likelihood of correct classification given the image $\mathbf{x}$.
The joint probability of the class $c$ and features $\mathbf{f}$ is modeled as:
$$P(c, \mathbf{f} | \mathbf{x}; \theta, \phi) = P(c | \mathbf{f}, \mathbf{x}; \phi) \times P(\mathbf{f} | \mathbf{x}; \theta)$$
Because the feature extraction function $\mathbf{f} = e(\mathbf{x})$ is deterministic, the model simplifies the marginalization to focus on the classification probability $P(c | \mathbf{x}; \theta, \phi)$. The training objective is to minimize the negative log-likelihood (cross-entropy loss):
$$\mathcal{L}_{\text{loss}}(\theta, \phi) = -\sum_{i=1}^{N} \log P(\hat{c}^i | \mathbf{x}^i; \theta, \phi)$$
By minimizing this loss, the model simultaneously learns to select the most relevant radiomic features (via $\theta$) and classify the injury (via $\phi$). The "hard selection" at inference (binarizing probabilities with a threshold $T$) is a heuristic that bridges the gap between the soft training weights and the need for a discrete set of features for clinical interpretability. The inclusion of the healthy persona effectively doubles the feature pool to $F = 6mP$, allowing the model to compare the patient's actual state against their own reconstructed healthy baseline, which is a clever way to isolate pathological changes.
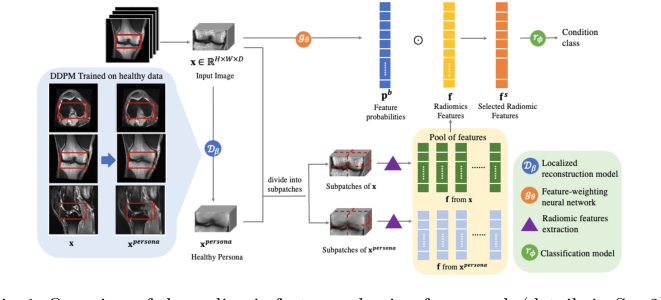 Figure 1. Overview of the radiomic feature selection framework (details in Sec.2)
Figure 1. Overview of the radiomic feature selection framework (details in Sec.2)
The framework successfully demonstrates that by combining generative AI with traditional statistical models, we can achieve high-performance diagnostics without sacrificing the "human-explainable" nature of the features. It is a robust approach to medical image analysis that feels like a significant step forward in trustable AI.
Problem Definition & Constraints
Core Problem Formulation & The Dilemma
The Starting Point (Input):
The current state of medical imaging diagnosis for knee injuries is dominated by two competing paradigms: "hand-crafted" radiomic features (interpretable but weak) and end-to-end Deep Learning (powerful but opaque).
The Desired Endpoint (Goal):
The authors aim to bridge this gap by creating a framework that achieves the high diagnostic performance of deep learning while retaining the human-readable interpretability of classical radiomics.
The Dilemma & Constraints:
1. The Interpretability Wall: Standard DL models are opaque.
2. The Feature Selection Bottleneck: Using all radiomic features leads to the "curse of dimensionality."
3. The Lack of Baseline: Without a patient-specific healthy baseline, it is hard to quantify exactly how much a tissue has deviated from its healthy state.
4. Computational & Data Constraints: The authors had to overcome the difficulty of training a generative model (DDPM) to accurately reconstruct healthy tissue without introducing artifacts that would bias the subsequent radiomic analysis.
Why This Approach
The authors identified a fundamental bottleneck: the "black box" nature of end-to-end DL. They developed a framework that uses DL to optimize the selection of radiomic features.
The Logic of the Approach
- The Feature-Weighting Network: A 3D-ResNet-18 acts as a gating mechanism, predicting probabilities $p = \{p_i\}_{i=1}^F$ for a pool of radiomic features.
- The Healthy Persona: They used a 3D Denoising Diffusion Probabilistic Model (DDPM) to reconstruct a "healthy persona" of the patient's knee. By extracting features from both the pathological image and its healthy synthetic counterpart, they created a differential feature set that highlights abnormalities more effectively.
Comparative Superiority
- Structural Advantage: This method reduces the input to a set of interpretable, patch-based radiomic features, maintaining a linear, human-explainable decision boundary.
- Generative Augmentation: The DDPM provides an explicit, patient-specific reference point, reducing the burden on the classifier to infer the baseline anatomy.
Mathematical & Logical Mechanism
The core of the training process is the minimization of the negative log-likelihood:
$$\mathcal{L}_{\text{loss}}(\theta, \phi) = -\sum_{i=1}^{N} \log P(\hat{c}^i | \mathbf{x}^i; \theta, \phi)$$
Step-by-step Flow
- Input: A raw MRI patch $\mathbf{x}$ enters the system.
- Persona Generation: A 3D DDPM takes the masked pathological image and "fills in the blanks" to generate a healthy persona.
- Feature Extraction: The system extracts radiomic features from both the original patch and the healthy persona.
- Weighting: The feature-weighting network $g_\theta(\mathbf{x})$ assigns a probability $p_i$ to each feature.
- Classification: The weighted features $\mathbf{f}^w = \{p_i f_i\}$ are fed into the logistic regression model $r_\phi$.
Results, Limitations & Conclusion
Evidence of Success
The authors defeated several baseline models, including MRNet, ELNet, and SKID.
* Performance: Their method achieved an accuracy of 0.90 for general abnormalities and 0.82 for meniscus tears.
* Ablation Proof: Removing the "healthy persona" or the "feature-weighting network" resulted in statistically significant drops in performance.
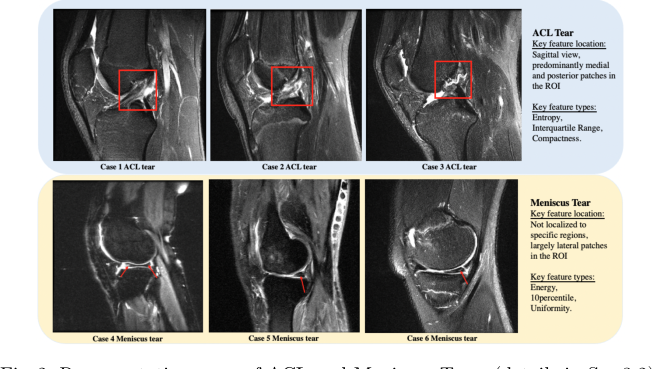 Figure 2. Representative cases of ACL and Meniscus Tears (details in Sec.3.2)
Figure 2. Representative cases of ACL and Meniscus Tears (details in Sec.3.2)
Future Discussion Topics
- Generalization to Other Pathologies: Could this "healthy persona" approach be applied to more complex, heterogeneous diseases like tumors?
- Uncertainty Quantification: Future iterations could explore probabilistic thresholding to provide radiologists with a "confidence score."
- Computational Efficiency: Can we develop "distilled" versions of these generative models that provide a healthy persona in near real-time?
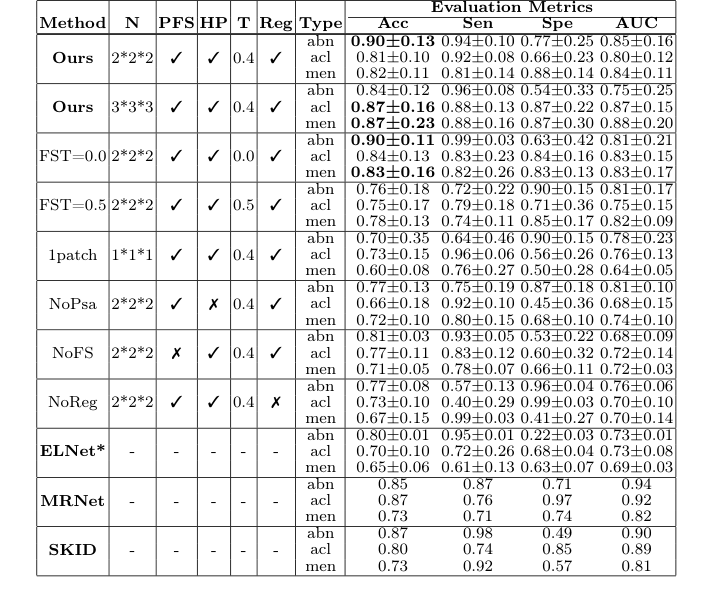 Table 1. Comparison of different configurations and methods. N: Number of subpatches; PFS: Patient-specific features selection; HP: Healthy persona; T: Feature selection threshold; Reg: Registration in preprocessing. * denotes repro- duced results
Table 1. Comparison of different configurations and methods. N: Number of subpatches; PFS: Patient-specific features selection; HP: Healthy persona; T: Feature selection threshold; Reg: Registration in preprocessing. * denotes repro- duced results
