Multifunctional fiber-optic theranostic probe for closed-loop tumor photothermal therapy
The combination of optical fiber and phototheranostic agents has emerged as a promising strategy to address the challenges of limited light penetration depth and systemic toxicity of nanomaterials.
Background & Academic Lineage
The Origin & Academic Lineage
The problem addressed in this paper originates from the critical global health challenge posed by cancer, with millions of new cases and deaths annually. This dire situation has spurred extensive research into developing more accurate and effective diagnostic and therapeutic methods. Within this context, the concept of "theranostics" emerged, aiming to integrate diagnostic and therapeutic functions into a single, spatially co-localized platform to enable immediate, targeted therapy and real-time monitoring.
Specifically, photo-theranostics, which leverages light for both diagnosis and therapy, gained significant attention due to its high specificity, spatiotemporal control, and non-ionizing nature. However, its clinical translation faced two primary hurdles: the inherently limited penetration depth of light in biological tissues (typically less than 10 mm due to scattering and absorption) and the systemic toxicity caused by the non-specific accumulation of nanomaterials used as theranostic agents in healthy tissues.
The combination of optical fibers with phototheranostic agents was identified as a promising strategy to overcome these limitations. Optical fibers facilitate efficient light transmission to deep-seated tumors and allow for localized confinement of therapeutic agents, thereby mitigating systemic toxicity. Despite these advantages, previous fiber-optic probe designs suffered from significant "pain points." Most existing approaches were limited to single-function-per-fiber implementations or experienced inter-functional crosstalk due to spectral overlap among the agents. This often necessitated multi-fiber configurations, leading to increased device rigidity, larger dimensions, more invasive procedures, and a lack of real-time therapeutic feedback. These issues collectively increased the risk of tissue damage and post-treatment inflammation, hindering widespread clinical adoption. This paper directly addresses these fundamental limitations by proposing a novel single-fiber multifunctional integration scheme, inspired by wavelength division multiplexing (WDM) technology, to enable closed-loop tumor photothermal therapy with real-time feedback.
Intuitive Domain Terms
- Theranostics: Imagine a highly advanced medical tool that acts like a "diagnose-and-treat" superhero. It can instantly tell a doctor what's wrong inside your body and then immediately deliver the right treatment to that exact spot, all in one go. It's a combination of diagnosis and therapy.
- Photothermal Therapy (PTT): Think of it as using a tiny, super-focused laser beam to gently heat up and destroy unwanted cells, much like how a magnifying glass can concentrate sunlight to burn a small spot on a leaf. The "photo" part means light is used, and "thermal" means heat is generated.
- Wavelength Division Multiplexing (WDM): Picture a single, very busy highway, but instead of cars, it carries different types of information, each traveling on a distinct "color" of light. This allows many different signals to travel simultaneously on the same fiber without interfering with each other, making the highway much more efficient.
- Tumor Microenvironment (TME): This refers to the immediate surroundings of a tumor, including all the cells, blood vessels, and chemical signals that support its growth. It's like the specific "neighborhood" a tumor creates for itself, which can be quite different from healthy tissue, often being more acidic.
- Ratiometric Fluorescent Probes: Consider a smart light sensor that doesn't just measure how bright one color of light is, but instead measures the brightness of two different colors and compares their ratio. This makes the measurement much more reliable and accurate, even if the overall brightness changes due to external factors, similar to how comparing two numbers can give a more stable result than relying on just one.
Notation Table
| Notation | Description |
|---|---|
| $I_{450}/I_{405}$ | Ratio of fluorescence intensities at 450 nm and 405 nm, used for pH sensing. |
| $R$ | pH resolution, indicating the smallest change in pH that can be reliably detected. |
| $s$ | Sensitivity, representing the slope of the pH calibration curve. |
| $T_1$ | Response time for pH or temperature sensing when the value increases. |
| $T_2$ | Response time for pH or temperature sensing when the value decreases. |
| $I_{618}/I_{546}$ | Ratio of fluorescence intensities at 618 nm and 546 nm, used for temperature sensing. |
| $p$ | p-value, a statistical measure used to determine the significance of experimental results. |
| $V/V_0$ | Normalized tumor volume, representing the tumor volume relative to its initial volume. |
| $\sigma$ | Standard deviation, a measure of the amount of variation or dispersion in a set of values. |
Problem Definition & Constraints
Core Problem Formulation & The Dilemma
The fundamental problem this paper addresses stems from the critical need for effective and minimally invasive cancer theranostics – a combined diagnostic and therapeutic approach. Current methods face significant limitations that hinder their clinical translation.
The Input/Current State can be characterized by several shortcomings:
1. Limited Light Penetration & Systemic Toxicity: Traditional phototheranostic agents, often nanomaterial-based, suffer from inherently limited light penetration depth (typically less than 10 mm) into tissues due to scattering and absorption. Furthermore, their non-specific accumulation in normal tissues and organs leads to systemic toxicity.
2. Underrated Multiplexing Potential of Fiber Optics: While optical fibers offer advantages like deep-seated tumor access and localized confinement of agents, their potential for integrating multiple functions on a single platform has been largely underutilized.
3. Inter-functional Crosstalk & Device Complexity: Previous fiber-optic probes were either limited to single-function-per-fiber implementations or, when attempting to integrate multiple functions, suffered from inter-functional crosstalk. This crosstalk primarily arose from spectral overlap in the absorption or emission bands of different functional reagents. To circumvent this, researchers often resorted to multi-fiber configurations, which inevitably increased device rigidity and dimensions, making them less compatible with minimally invasive interventional techniques and elevating risks of tissue damage and post-treatment inflammation.
4. Lack of Real-time Therapeutic Feedback: Existing methods often lack real-time monitoring capabilities for therapeutic dose and efficacy, preventing closed-loop control and personalized precision medicine.
The Desired Endpoint (Output/Goal State) is a single, compact, multifunctional fiber-optic theranostic probe capable of:
1. Closed-loop Tumor Photothermal Therapy (PTT): This involves integrating diagnostic and therapeutic functions on a single platform for real-time feedback.
2. Multi-parameter Monitoring: The probe should enable simultaneous monitoring of key tumor microenvironment (TME) parameters, specifically pH and temperature.
3. Minimally Invasive Intervention: The device should be compact (e.g., 440 µm diameter, as achieved by the authors) to access tumor lesions via interventional procedures, avoiding enlarged incisions and repeated invasive procedures.
4. Pre-treatment Tumor Identification: Accurately identifying tumor edges by revealing the tumor pH gradient.
5. Intra-treatment Thermal Dose Control: Precisely controlling the thermal dose during PTT by simultaneously monitoring temperature.
6. Post-treatment Efficacy Assessment: Rapidly assessing treatment efficacy by monitoring the reversal of the acidic TME.
7. Overcoming Light Penetration & Toxicity: Addressing the limitations of light penetration depth and systemic toxicity associated with nanomaterial-based strategies.
The exact missing link or mathematical gap this paper attempts to bridge is the development of a robust, single-fiber platform that can simultaneously perform multiple theranostic functions (pH sensing, temperature sensing, and photothermal therapy) without inter-functional crosstalk, while providing real-time feedback. The core mathematical/design challenge is to select and co-immobilize multiple functional agents (pH indicator, temperature indicator, photothermal agent) such that their excitation and emission spectra are sufficiently separated to allow for wavelength division multiplexing (WDM) on a single fiber. This enables distinct functions to be activated on-demand by specific wavelengths, effectively eliminating the spectral overlap problem that plagued previous single-fiber multiplexing attempts. The paper's solution involves carefully choosing HPTS-IP, LnMOF, and ICG, whose absorption bands (405/450 nm for HPTS-IP, 295 nm for LnMOF, 790 nm for ICG) are non-overlapping, thus enabling WDM.
The painful trade-off or dilemma that has trapped previous researchers is the inherent conflict between functional multiplexing and device simplicity/performance. To achieve multiple functions, one either had to:
* Increase device complexity: Use multiple fibers, leading to bulky, rigid devices unsuitable for minimally invasive procedures and increasing patient risk.
* Compromise functional integrity: Attempt to integrate multiple agents on a single fiber, only to encounter severe inter-functional crosstalk due to spectral overlap, rendering the multi-functionality unreliable or inaccurate. This meant that improving one aspect (e.g., adding more functions) directly broke another (e.g., signal integrity or device size).
Constraints & Failure Modes
The problem of developing a multifunctional fiber-optic theranostic probe is insanely difficult due to several harsh, realistic walls the authors hit:
-
Physical Constraints:
- Limited Light Penetration Depth: Light's inherent scattering and absorption in biological tissues severely limit its penetration, typically to less than 10 mm. This makes treating deep-seated tumors challenging for photothermal therapies.
- Systemic Toxicity: Non-specific accumulation of theranostic nanomaterials in normal tissues and organs leads to undesirable systemic toxicity, a major hurdle for clinical translation.
- Device Rigidity and Dimensions: For minimally invasive procedures, the probe must be flexible and compact. Multi-fiber designs increase rigidity and dimensions, making them incompatible with interventional techniques and increasing the risk of tissue damage.
- Tapered Fiber Design Limitations: The geometry of the tapered optical fiber is critical. Excessively short tapers risk light leakage and degrade the signal-to-noise ratio (SNR), while overlong tapers are unsuitable for localized sensing and therapy. The tip diameter affects both evanescent field strength (and thus fluorescence excitation/collection efficiency) and the available surface area for indicator immobilization, requiring careful optimization (e.g., 100 µm was found optimal).
- Hydrogel Film Integrity: The sol-gel matrix used to encapsulate the functional agents must be stable and biocompatible. Pure tetraethyl orthosilicate (TEOS) films are prone to severe cracking, leading to low film uniformity and high leaching rates, which compromises sensor stability and biocompatibility. The addition of co-precursors like GLYMO and surfactants like Triton X-100 is necessary to mitigate this.
-
Data-driven & Performance Constraints:
- Inter-functional Crosstalk: The primary challenge for multiplexing on a single fiber is preventing spectral overlap between the absorption and emission bands of different functional reagents (pH indicator, temperature indicator, photothermal agent). This overlap would lead to inaccurate readings and an inability to distinguish between signals from different functions.
- Real-time Latency Requirements: For closed-loop therapy and real-time monitoring, the sensors must have sufficiently fast response times. The paper reports pH sensing response times of 3.2 s (increasing pH) and 12.8 s (decreasing pH), and temperature sensing response times of 2.0 s (increasing temperature) and 0.6 s (decreasing temperature), which are deemed acceptable for in vivo monitoring.
- Sensor Stability and Reliability: The functional agents must remain stable and not leach significantly from the fiber surface over time in a physiological environment. Leaching tests showed minimal decrease in fluorescence intensity (1.6% for HPTS-IP, 3.5% for LnMOF over 72h), indicating good stability.
- Interference from Physiological Factors: The sensing performance must be robust against various interfering substances present in complex physiological environments, as well as photobleaching, indicator concentration variations, and changes in fiber morphology. The use of ratiometric fluorescent probes and careful agent selection helps mitigate these issues.
- Tissue Autofluorescence: Endogenous fluorescence from tissues can interfere with the probe's signals, degrading sensing accuracy and specificity. The shallow penetration depth of the evanescent field, where indicator concentration is high, helps maintain high SNR.
- Precise Thermal Dose Control: During photothermal therapy, delivering an effective thermal dose without causing excessive heating to adjacent normal tissues is critical. Overheating can lead to tissue damage, vascular collapse, and impaired blood perfusion. This necessitates accurate, real-time temperature monitoring.
Why This Approach
The Inevitability of the Choice
The authors' decision to pursue a single-fiber multifunctional integration strategy, leveraging wavelength division multiplexing (WDM) technology, was not merely an improvement but an inevitable choice driven by the inherent limitations of existing approaches. The paper clearly identifies the exact moment traditional "state-of-the-art" (SOTA) methods fell short: "Despite recent advances, the multiplexing potential of fiber-optic probes remains underrated. Current research remains limited to single-function-per-fiber implementations or suffers from inter-functional crosstalk, which primarily arises from spectral overlap in the absorption or emission bands among the functional reagents used." This spectral overlap issue meant that achieving multi-parameter monitoring or integrated theranostics often necessitated multi-fiber configurations. Such setups, however, "inevitably increases device rigidity and dimensions, decreasing compatibility for interventional techniques while elevating risks of tissue damage and post-treatment inflammation." Given these critical drawbacks—lack of real-time feedback, invasiveness, and functional crosstalk—a paradigm shift was required. The WDM-based single-fiber approach emerged as the only viable solution to overcome these fundamental challenges by enabling multiple functions on a single, compact platform without spectral interference.
Comparative Superiority
Beyond simple performance metrics, this method offers profound qualitative superiority, primarily through its structural design. The core advantage lies in its ability to integrate multiple functions (pH sensing, temperature monitoring, and photothermal therapy) onto a single optical fiber using wavelength division multiplexing. This structural innovation inherently minimizes inter-functional crosstalk, a significant problem in previous multi-agent systems where spectral overlap degraded signal integrity. By carefully selecting agents (HPTS-IP, LnMOF, ICG) with "non-overlapping absorption bands," the probe ensures accurate and reliable measurement of each parameter.
Furthermore, the chosen ratiometric fluorescent probes for pH and temperature sensing provide a crucial qualitative advantage. This ratiometric detection strategy "mitigates interference from photobleaching, indicator concentration variations, and fiber morphology changes on the sensing results, facilitating the acquisition of accurate and reliable signals." This is a significant improvement over intensity-based sensing, which is highly susceptible to noise and environmental fluctuations. The compact, single-fiber design also drastically reduces the physical footprint and rigidity compared to multi-fiber bundles, making it overwhelmingly superior for minimally invasive interventions. It also localizes the theranostic agents, effectively mitigating systemic toxicity associated with free nanomaterials.
Alignment with Constraints
The chosen method perfectly aligns with the stringent constraints of advanced theranostic applications.
- Minimally Invasive: The use of a single, flexible optical fiber (diameter = 440 µm, tip diameter = 100 µm) allows for "accessing tumor lesions via interventional procedures," drastically reducing the invasiveness compared to multi-fiber or larger probe designs. This directly addresses the constraint of minimizing tissue damage and post-treatment inflammation.
- Real-time Feedback & Closed-Loop Therapy: The integration of pH and temperature indicators with a photothermal agent on a single platform enables "closed-loop tumor photothermal therapy with real-time feedback." This means the probe can perform pre-treatment tumor identification (via pH gradient), intra-treatment dose control (via temperature monitoring during PTT), and post-treatment efficacy assessment (via TME pH reversal), fulfilling the critical need for dynamic, adaptive treatment.
- Multi-parameter Monitoring: The wavelength division multiplexing scheme allows for simultaneous monitoring of pH and temperature, alongside photothermal therapy, on a single fiber. This directly meets the requirement for comprehensive multi-parameter assessment in complex biological enviornments.
- Reduced Systemic Toxicity & Deep Tissue Access: By immobilizing the phototheranostic agents onto the optical fiber surface, the approach "effectively mitigates off-target toxicity through localized confinement." Additionally, optical fibers facilitate "end-to-end light transmission with minimal loss," enabling sensing and treatment of "deep-seated tumors," thereby overcoming the limited light penetration depth of traditional phototheranostic agents.
Rejection of Alternatives
The paper implicitly and explicitly rejects several alternative approaches based on their fundamental limitations for the specific problem of closed-loop, multifunctional tumor theranostic.
- Single-function-per-fiber implementations: These were deemed insufficient becuase they would require multiple fibers for multi-parameter monitoring and therapy, leading to "enlarged incisions, repeated invasive procedures," and increased device rigidity. The goal was integrated theranostics, not fragmented functionalities.
- Multi-fiber configurations: While offering multiplexing, these were rejected due to their inherent physical limitations, such as "increased device rigidity and dimensions," which compromise compatibility with minimally invasive interventional techniques and elevate risks of tissue damage.
- Approaches suffering from inter-functional crosstalk: Methods where functional reagents have "spectral overlap in the absorption or emission bands" were explicitly identified as problematic. The authors' careful selection of agents with non-overlapping bands and the WDM strategy directly addresses this failure point, ensuring signal integrity.
- Nanomaterial-dependent strategies (without fiber integration): The conclusion highlights that compared to "nanomaterial-dependent strategies," this fiber-optic approach "effectively addresses limitations of light penetration depth and systemic toxicity." This implies that relying solely on free-floating or non-fiber-integrated nanomaterials would fail to provide the necessary deep tissue access and localized, non-toxic delivery required for effective tumor photothermal therapy.
Mathematical & Logical Mechanism
The Master Equation
At the heart of this theranostic probe's sensing capabilities lies the Boltzmann sigmoid model, which serves as the fundamental transformation logic for converting ratiometric fluorescence signals into meaningful physiological parameters like pH and temperature. While the photothermal therapy component involves direct light-to-heat conversion, its precise control and monitoring are entirely dependent on the accurate readings provided by these sensing mechanisms.
The general form of the Boltzmann equation, as employed in this paper for both pH and temperature sensing, can be expressed as:
$$ y = C_1 - \frac{C_2}{1 + e^{(x - C_3)/C_4}} $$
Where:
- For pH sensing (derived from Fig. 2f):
$$
R_{pH} = 0.97 - \frac{0.78}{1 + e^{(pH - 7.07)/0.87}}
$$
Here, $y$ is the fluorescence intensity ratio $R_{pH} = I_{520}(450nm) / I_{520}(405nm)$, and $x$ is the pH value.
- For temperature sensing (derived from Fig. 3g):
$$ R_{Temp} = 1.55 - \frac{0.81}{1 + e^{(Temp - 65.74)/10.71}} $$
Here, $y$ is the fluorescence intensity ratio $R_{Temp} = I_{618} / I_{546}$, and $x$ is the temperature value in degrees Celsius.
These equations are the core mathematical engines that enable the probe to quantitatively interpret the optical signals it receives, providing the crucial feedback for closed-loop theranostics.
Term-by-Term Autopsy
Let's dissect the general Boltzmann equation $y = C_1 - \frac{C_2}{1 + e^{(x - C_3)/C_4}}$ to understand the role of each component:
-
$y$ (Fluorescence Intensity Ratio, $R_{pH}$ or $R_{Temp}$)
- Mathematical Definition: This is the dependent variable, representing a dimensionless ratio of fluorescence intensities. For pH sensing, it's the ratio of the emission intensity at 520 nm when excited at 450 nm to that when excited at 405 nm ($I_{520}(450nm) / I_{520}(405nm)$). For temperature sensing, it's the ratio of emission intensities at 618 nm to 546 nm ($I_{618} / I_{546}$) under 295 nm excitation.
- Physical/Logical Role: This term is the direct output of the optical measurement system. By using a ratio, the authors cleverly mitigate common sources of error such as photobleaching, variations in indicator concentration, and changes in fiber morphology. This normalization makes the signal robust and reliable, acting as the primary observable that changes predictably with the target parameter.
- Why Ratio/Subtraction/Division: The ratio is a standard technique in ratiometric sensing to self-reference and cancel out common-mode noise. The overall sigmoidal shape, involving subtraction and division, naturally arises from chemical or physical processes where an indicator transitions between two states (e.g., protonated/deprotonated, or different energy transfer states) in response to a continuous variable, exhibiting saturation at both extremes.
-
$x$ (Physiological Parameter, $pH$ or $Temp$)
- Mathematical Definition: This is the independent variable, representing the environmental condition being measured (pH or temperature in °C).
- Physical/Logical Role: This is the actual physical quantity that the theranostic probe is designed to detect and monitor. It's the "truth" value that the system aims to determine.
- Why Independent Variable: It's the cause, while $y$ is the effect. The calibration process establishes the functional relationship between this input and the measured output ratio.
-
$C_1$ (Upper Asymptote)
- Mathematical Definition: A constant coefficient representing the upper plateau of the sigmoidal curve as $x$ approaches positive infinity (for this specific form of decreasing sigmoid).
- Physical/Logical Role: For pH, $C_1 = 0.97$ represents the maximum fluorescence ratio achieved when the HPTS-IP indicator is predominantly in one of its protonation states (e.g., fully deprotonated at high pH). For temperature, $C_1 = 1.55$ represents the maximum ratio for LnMOF at high temperatures. It defines one extreme of the sensor's dynamic range, indicating a saturation point where further increases in $x$ no longer significantly alter the indicator's state.
- Why Constant: It's an asymptotic value, reflecting the maximum possible response of the indicator under extreme conditions, where the equilibrium has shifted almost entirely to one side.
-
$C_2$ (Range Scaling Factor)
- Mathematical Definition: A constant coefficient that scales the magnitude of the sigmoidal transition. It represents the total change in $y$ from its lower asymptote to its upper asymptote.
- Physical/Logical Role: For pH, $C_2 = 0.78$ dictates the overall span of the fluorescence ratio change as pH varies across the sensitive range. For temperature, $C_2 = 0.81$ does the same for temperature. This term essentially defines the "height" of the sigmoidal step, indicating the total dynamic range of the sensor's response.
- Why Constant: It's a characteristic property of the indicator and its interaction with the environment, reflecting the maximum possible change in the ratiometric signal.
-
$C_3$ (Midpoint or Center of Transition)
- Mathematical Definition: A constant coefficient representing the $x$-value at which the response is halfway between its asymptotic limits (or where the slope is maximal).
- Physical/Logical Role: For pH, $C_3 = 7.07$ indicates the pH value where the HPTS-IP indicator is most sensitive to changes, often corresponding to its pKa. For temperature, $C_3 = 65.74$ °C marks the temperature where the LnMOF's energy transfer mechanism exhibits its steepest change. This parameter is crucial for defining the working range and optimal sensitivity of the sensor.
- Why Constant: It's an intrinsic property of the chemical or physical process being monitored by the indicator.
-
$C_4$ (Slope Parameter)
- Mathematical Definition: A constant coefficient that is inversely proportional to the maximum slope of the sigmoidal curve. A smaller absolute value of $C_4$ indicates a steeper curve.
- Physical/Logical Role: For pH, $C_4 = 0.87$ quantifies the steepness of the pH response curve. For temperature, $C_4 = 10.71$ does the same for temperature. This parameter directly relates to the sensor's sensitivity: a smaller $C_4$ means a more rapid change in the fluorescence ratio for a given change in $x$ around the midpoint $C_3$.
- Why Constant: It reflects the inherent responsiveness and cooperativity of the indicator's transition.
-
$e$ (Euler's Number)
- Mathematical Definition: A mathematical constant, the base of the natural logarithm, approximately 2.71828.
- Physical/Logical Role: It's fundamental to exponential functions, which are used here to model the sigmoidal transition. Such transitions often arise from statistical mechanics or chemical equilibrium equations (like the Henderson-Hasselbalch equation for pH, or Boltzmann distributions for energy states), where the probability of a state changes exponentially with the driving force.
- Why Exponential: The exponential term naturally describes the non-linear, saturating behavior observed in many physical and chemical systems as they approach equilibrium.
-
Exponent $(x - C_3)/C_4$
- Mathematical Definition: The argument of the exponential function.
- Physical/Logical Role: This term normalizes the difference between the current input $x$ and the midpoint $C_3$ by the slope parameter $C_4$. It effectively scales the input to fit the exponential decay or growth, determining how far the system is from its half-transition point.
-
Division by $(1 + e^{(x - C_3)/C_4})$
- Mathematical Definition: This specific form is the core of the sigmoidal shape, often referred to as the logistic function or Fermi-Dirac distribution.
- Physical/Logical Role: This term ensures that the output $y$ smoothly transitions between the two asymptotes as $x$ changes. It models the gradual shift in the indicator's state from one extreme to the other.
-
Subtraction $C_1 - \frac{C_2}{...}$
- Mathematical Definition: This operation combines the upper asymptote with the scaled sigmoidal term.
- Physical/Logical Role: For the specific form used in the paper, this subtraction creates a decreasing sigmoid if $C_2$ is positive, meaning the ratio $y$ starts high and decreases towards $C_1 - C_2$ as $x$ increases. However, the graphs (Fig. 2f and 3g) show increasing ratios with increasing pH/Temp. This implies that the authors' fitted equation $y = C_1 - \frac{C_2}{1 + e^{(x - C_3)/C_4}}$ is actually an increasing sigmoid where $C_1$ is the upper asymptote and $C_1 - C_2$ is the lower asymptote. As $x \to -\infty$, $e^{(x - C_3)/C_4} \to 0$, so $y \to C_1 - C_2$. As $x \to +\infty$, $e^{(x - C_3)/C_4} \to \infty$, so $y \to C_1$. This is consistent with the figures. The subtraction is integral to defining the curve's orientation and range.
Step-by-Step Flow
Imagine the theranostic probe as a sophisticated, miniature assembly line for data processing, where each step transforms raw optical signals into actionable physiological insights.
- Light Input & Wavelength Selection: The process begins with light from a tunable source entering the Y-type optical fiber. Depending on the desired measurement, a specific wavelength is selected, much like choosing a tool from a toolbox. For pH sensing, the system rapidly alternates between 405 nm and 450 nm excitation light. For temperature sensing, a 295 nm excitation wavelength is used. This wavelength division multiplexing (WDM) is the initial "switch" that determines the probe's function.
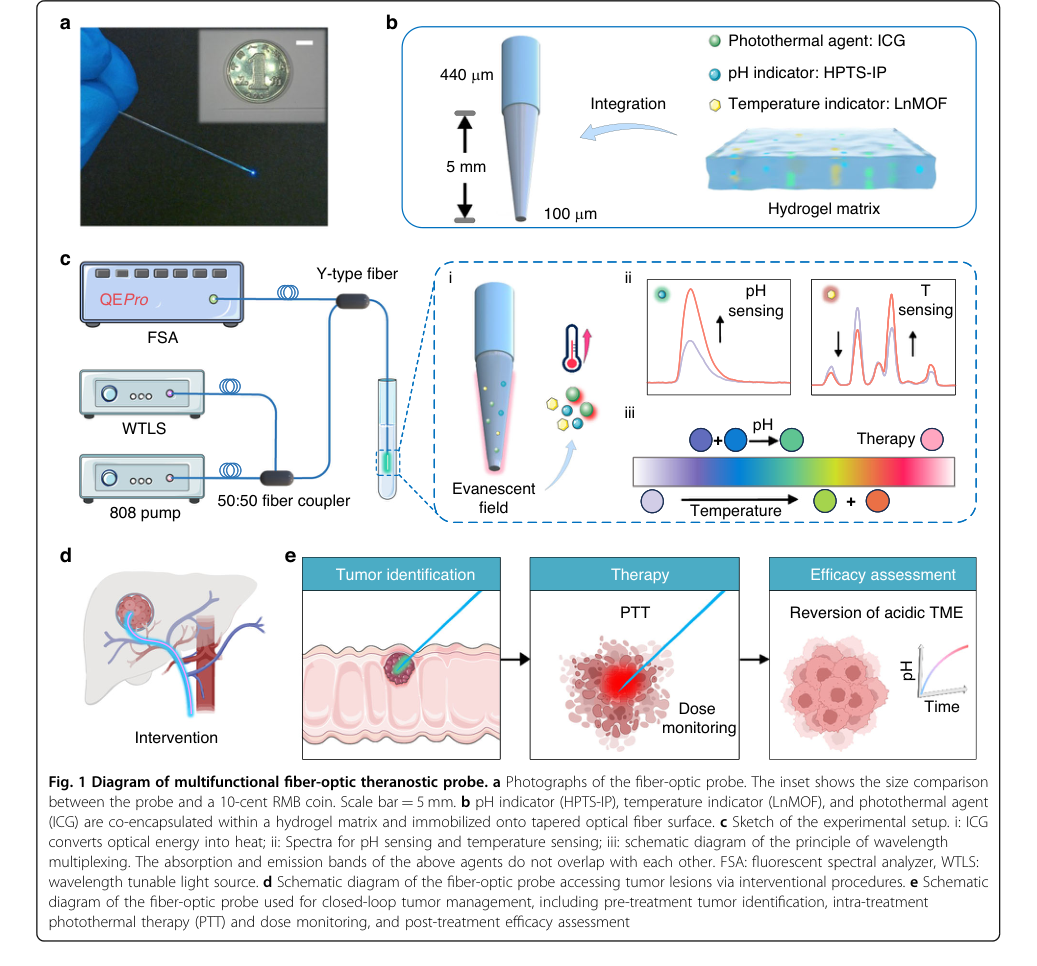 Figure 1. Diagram of multifunctional fiber-optic theranostic probe. a Photographs of the fiber-optic probe. The inset shows the size comparison between the probe and a 10-cent RMB coin. Scale bar = 5 mm. b pH indicator (HPTS-IP), temperature indicator (LnMOF), and photothermal agent (ICG) are co-encapsulated within a hydrogel matrix and immobilized onto tapered optical fiber surface. c Sketch of the experimental setup. i: ICG converts optical energy into heat; ii: Spectra for pH sensing and temperature sensing; iii: schematic diagram of the principle of wavelength multiplexing. The absorption and emission bands of the above agents do not overlap with each other. FSA: fluorescent spectral analyzer, WTLS: wavelength tunable light source. d Schematic diagram of the fiber-optic probe accessing tumor lesions via interventional procedures. e Schematic diagram of the fiber-optic probe used for closed-loop tumor management, including pre-treatment tumor identification, intra-treatment photothermal therapy (PTT) and dose monitoring, and post-treatment efficacy assessment
Figure 1. Diagram of multifunctional fiber-optic theranostic probe. a Photographs of the fiber-optic probe. The inset shows the size comparison between the probe and a 10-cent RMB coin. Scale bar = 5 mm. b pH indicator (HPTS-IP), temperature indicator (LnMOF), and photothermal agent (ICG) are co-encapsulated within a hydrogel matrix and immobilized onto tapered optical fiber surface. c Sketch of the experimental setup. i: ICG converts optical energy into heat; ii: Spectra for pH sensing and temperature sensing; iii: schematic diagram of the principle of wavelength multiplexing. The absorption and emission bands of the above agents do not overlap with each other. FSA: fluorescent spectral analyzer, WTLS: wavelength tunable light source. d Schematic diagram of the fiber-optic probe accessing tumor lesions via interventional procedures. e Schematic diagram of the fiber-optic probe used for closed-loop tumor management, including pre-treatment tumor identification, intra-treatment photothermal therapy (PTT) and dose monitoring, and post-treatment efficacy assessment
-
Evanescent Field Interaction: The selected light travels down the fiber to its tapered tip, where it escapes into the surrounding hydrogel matrix as an "evanescent field." This field acts as a microscopic net, interacting directly with the co-immobilized indicator molecules (HPTS-IP for pH, LnMOF for temperature). The indicators absorb this light, becoming energized.
-
Fluorescence Emission: Upon excitation, the energized indicator molecules emit fluorescence. The characteristics of this emitted light are sensitive to the local environment. For pH, HPTS-IP emits fluorescence at 520 nm, but its intensity at this wavelength varies depending on whether it was excited at 405 nm or 450 nm, reflecting the local protonation state. For temperature, LnMOF emits fluorescence at two distinct wavelengths, 546 nm and 618 nm, with their relative intensities changing based on the local temperature due to energy transfer dynamics.
-
Signal Collection: The emitted fluorescence is then efficiently collected back into the same optical fiber, like a vacuum cleaner sucking up the light, and guided away from the tissue.
-
Spectral Analysis: The collected light travels to a Fluorescent Spectral Analyzer (FSA). This device acts as a "light sorter," measuring the precise intensity of the fluorescence at the relevant emission wavelengths. For pH, it measures $I_{520}(450nm)$ and $I_{520}(405nm)$. For temperature, it measures $I_{618}$ and $I_{546}$.
-
Ratiometric Calculation: A computational unit then calculates a dimensionless ratio from these raw intensity measurements. For pH, it computes $R_{pH} = I_{520}(450nm) / I_{520}(405nm)$. For temperature, it computes $R_{Temp} = I_{618} / I_{546}$. This ratio is the standardized "data point" that will be fed into the mathematical engine.
-
Boltzmann Model Transformation: This calculated ratio ($R_{pH}$ or $R_{Temp}$) is then input into the pre-calibrated Boltzmann equation. The equation, acting as a "decoder," performs a non-linear transformation, effectively inverting the calibration curve to yield the actual physiological parameter. For example, if $R_{pH}$ is fed into the pH Boltzmann equation, it outputs the corresponding pH value. Similarly, $R_{Temp}$ yields the temperature.
-
Real-time Feedback & Action: The derived pH and temperature values are displayed in real-time. These precise readings serve as critical feedback. During photothermal therapy (PTT), the temperature reading allows for "thermal dose control," enabling the operator to adjust the 808 nm pump laser power to maintain a target temperature (e.g., 65 °C at the fiber tip, ensuring 45 °C at the tumor edge). Post-treatment, pH monitoring assesses efficacy by tracking the reversal of the acidic tumor microenvironment. This closed-loop system ensures that the therapy is not only delivered but also continuously optimized based on the patient's real-time physiological state.
Optimization Dynamics
The "optimization dynamics" of this theranostic probe primarily reside in its initial calibration and its inherent design for robust, real-time feedback control, rather than continuous iterative learning in the machine learning sense.
-
Calibration as Initial "Learning":
- Data Acquisition: Before clinical application, the probe undergoes a rigorous calibration process. This involves exposing the sensor to a range of known pH values (e.g., pH 4.0 to 9.0) and temperatures (e.g., 30 °C to 100 °C) in controlled laboratory settings. At each known point, the corresponding ratiometric fluorescence signal is measured.
- Model Fitting: These collected data points (known parameter $x$ vs. measured ratio $y$) are then used to "train" or "fit" the Boltzmann sigmoid model. This fitting process is a form of non-linear regression, where the coefficients ($C_1, C_2, C_3, C_4$) of the Boltzmann equation are iteratively adjusted to minimize the difference (e.g., sum of squared errors) between the model's predicted ratio and the experimentally observed ratio for each known input. This is akin to navigating a "loss landscape" to find the optimal set of parameters that best describe the sensor's response curve. The paper reports high $R^2$ values (0.997 for pH, 0.997 for temperature), indicating an excellent fit and thus a well-defined loss landscape with a clear minimum.
- Parameter Fixation: Once these coefficients are determined, they are fixed. The sensor does not "learn" or update these parameters during its operational phase. This one-time calibration establishes the sensor's "knowledge" base.
-
Ratiometric Design for Robustness (Implicit Optimization):
- The choice of ratiometric sensing is a critical design "optimization." By taking a ratio of two fluorescence intensities (either from different excitation wavelengths for pH or different emission wavelengths for temperature), the system inherently cancels out many common noise sources. This includes fluctuations in excitation light intensity, photobleaching of the indicator, variations in indicator concentration, and even minor changes in the fiber's optical properties or coupling efficiency.
- This mechanism ensures that the signal-to-noise ratio (SNR) remains high and the sensor's output is stable and reliable, effectively "optimizing" the measurement quality in real-time without explicit algorithmic updates. The stability tests (Fig. 2g,h,i,j and Fig. 3j,k,l,n) confirm this robustness, showing minimal standard deviation and leaching.
-
Closed-Loop Feedback for Therapeutic Optimization:
- During actual photothermal therapy, the system operates as a closed-loop control mechanism. The sensor continuously provides real-time temperature and pH readings.
- These readings are not used to update the sensor's internal mathematical model, but rather to adjust the external therapeutic input. For instance, if the monitored temperature deviates from the target thermal dose, the pump laser power (the therapeutic input) is adjusted accordingly. This is a classic feedback loop: Sense (temperature/pH) $\rightarrow$ Evaluate (against target) $\rightarrow$ Act (adjust laser power).
- This iterative adjustment of the therapeutic input based on real-time sensor data is how the therapy itself is optimized to achieve desired outcomes (e.g., maintaining tumor temperature at 65 °C at the fiber tip to ensure 45 °C at the tumor edge) while minimizing side effects. The system converges towards the target therapeutic state through continuous monitoring and adjustment, making the treatment precise and adaptive.
In essence, the mathematical engine "learns" its characteristic response once during calibration, and then reliably applies this learned function. The overall system's "optimization" during therapy is achieved through a robust, real-time feedback loop that leverages the sensor's accurate and stable measurements to control the therapeutic intervention.
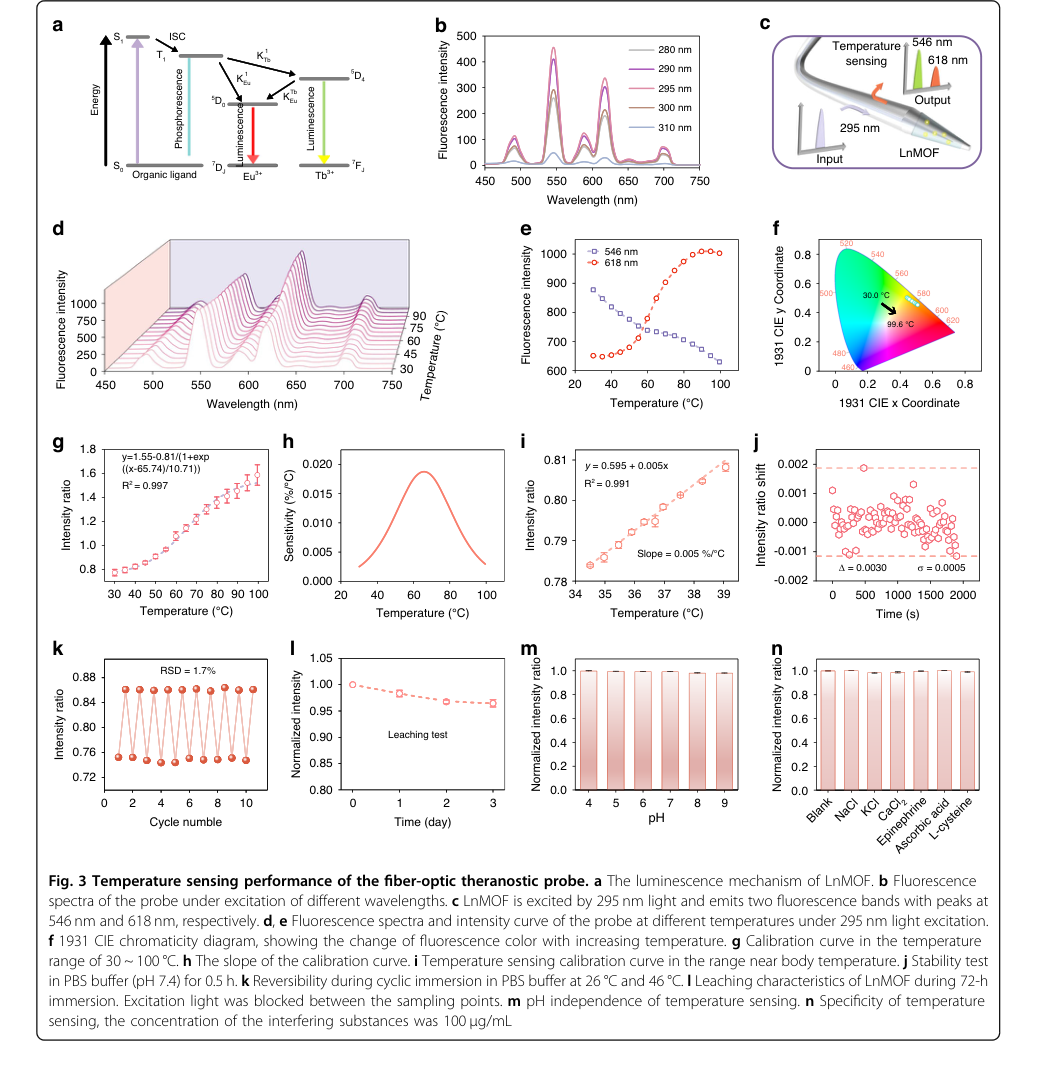 Figure 3. Temperature sensing performance of the fiber-optic theranostic probe. a The luminescence mechanism of LnMOF. b Fluorescence spectra of the probe under excitation of different wavelengths. c LnMOF is excited by 295 nm light and emits two fluorescence bands with peaks at 546 nm and 618 nm, respectively. d, e Fluorescence spectra and intensity curve of the probe at different temperatures under 295 nm light excitation. f 1931 CIE chromaticity diagram, showing the change of fluorescence color with increasing temperature. g Calibration curve in the temperature range of 30 ~ 100 °C. h The slope of the calibration curve. i Temperature sensing calibration curve in the range near body temperature. j Stability test in PBS buffer (pH 7.4) for 0.5 h. k Reversibility during cyclic immersion in PBS buffer at 26 °C and 46 °C. l Leaching characteristics of LnMOF during 72-h immersion. Excitation light was blocked between the sampling points. m pH independence of temperature sensing. n Specificity of temperature sensing, the concentration of the interfering substances was 100 μg/mL
Figure 3. Temperature sensing performance of the fiber-optic theranostic probe. a The luminescence mechanism of LnMOF. b Fluorescence spectra of the probe under excitation of different wavelengths. c LnMOF is excited by 295 nm light and emits two fluorescence bands with peaks at 546 nm and 618 nm, respectively. d, e Fluorescence spectra and intensity curve of the probe at different temperatures under 295 nm light excitation. f 1931 CIE chromaticity diagram, showing the change of fluorescence color with increasing temperature. g Calibration curve in the temperature range of 30 ~ 100 °C. h The slope of the calibration curve. i Temperature sensing calibration curve in the range near body temperature. j Stability test in PBS buffer (pH 7.4) for 0.5 h. k Reversibility during cyclic immersion in PBS buffer at 26 °C and 46 °C. l Leaching characteristics of LnMOF during 72-h immersion. Excitation light was blocked between the sampling points. m pH independence of temperature sensing. n Specificity of temperature sensing, the concentration of the interfering substances was 100 μg/mL
Results, Limitations & Conclusion
Experimental Design & Baselines
The researchers meticulously designed their experiments to rigorously validate the multifunctional capabilities and therapeutic efficacy of their novel fiber-optic theranostic probe. The core of their experimental architecture revolved around demonstrating the probe's ability to perform three distinct functions—pH sensing, temperature sensing, and photothermal therapy (PTT)—using a single optical fiber, leveraging wavelength division multiplexing (WDM) to prevent crosstalk.
For pH sensing, the probe, functionalized with HPTS-IP, was tested in phosphate-buffered saline (PBS) solutions across a broad pH range (4.0 to 9.0) at room temperature. Its stability was assessed by continuous spectral recording over 150 seconds in pH 7.4 PBS. Reversibility was proven by alternating immersion in pH 4.0 and 9.0 buffers over five cycles. Leaching characteristics were evaluated over 72 hours in pH 7.4 PBS. To confirm reliability in complex biological environments, the probe's specificity was tested against common interfering substances like NaCl, KCl, CaCl₂, epinephrine, ascorbic acid, and L-cysteine. Crucially, temperature crosstalk was assessed by varying temperatures from 26 to 41 °C while measuring pH.
Temperature sensing utilized LnMOF and was evaluated over a wide range (30-100 °C) and specifically within the physiologically relevant range (34.5-39 °C). Stability was tested for 0.5 hours in pH 7.4 PBS, and reversibility was demonstrated over 10 cycles by alternating immersion in 26 °C and 46 °C PBS. Leaching was monitored over 72 hours, and pH independence was confirmed by testing in buffers from pH 4.0 to 9.0. Specificity against interfering substances was also evaluated.
For photothermal conversion, the ICG-functionalized probe was subjected to in vitro tests using an 808 nm pump laser, measuring maximum temperatures at various pump powers (up to 267 mW). Photothermal stability was assessed over five heating/cooling cycles, and ICG leaching was monitored over 72 hours. The "victim" here was the typical requirement for high pump power in conventional nanomaterial-based PTT systems, which often exceed 1 W.
The ultimate validation came through in vivo experiments using subcutaneous colorectal cancer xenograft models in BALB/c nude mice. Tumors were allowed to reach approximately 100 mm³ before intervention.
- Tumor identification was rigorously tested by inserting the probe into tumor tissue, adjacent normal tissue, tumor surface, and normal tissue, comparing measured pH values.
- PTT and dose monitoring involved inserting the probe into the tumor center and actively maintaining the fiber temperature at 65 °C for 15 minutes, with thermal imaging confirming the tumor edge reached an effective therapeutic temperature of 45 °C.
- Efficacy assessment was multi-pronged: real-time pH monitoring in the tumor microenvironment (TME) post-treatment (30 minutes and 24 hours), and long-term monitoring of tumor volume and body weight every 3 days for 20 days. The "victims" in this context were untreated control group mice, whose tumors exhibited progressive growth, leading to euthanasia when volumes exceeded 2000 mm³.
- Biocompatibility was assessed in vitro by culturing HCT116 cells with probe leachate (24 hours) and performing CCK-8 assays and live-dead staining. In vivo, organ biocompatibility (heart, liver, spleen, lung, kidney) was evaluated via H&E staining 24 hours post-treatment in both implantation (fiber without laser) and treatment groups, compared to controls. Histological and immunohistochemical analyses (Caspase-3, HIF-1α, Ki67) of tumor tissues were performed 24 hours post-treatment, with sham-treated groups (fiber implantation without laser excitation) serving as a baseline for comparison.
What the Evidence Proves
The evidence presented in the paper definitively proves the successful development and functionality of a multifunctional fiber-optic theranostic probe, capable of closed-loop tumor photothermal therapy with real-time feedback.
The core mathematical claim of wavelength division multiplexing (WDM) for non-overlapping excitation bands was ruthlessly proven by the distinct absorption spectra of HPTS-IP (405 nm, 450 nm), LnMOF (295 nm), and ICG (790 nm), as shown in Fig. 2b and Fig. S7. This spectral separation is the undeniable evidence that the core mechanism for switching functions on demand works, effectively eliminating inter-functional crosstalk, a significant limitation of previous multi-agent systems.
For pH sensing, the probe demonstrated exceptional performance:
- It achieved a high resolution of 0.013 pH units within the linear range of 6.0 to 8.0, crucial for distinguishing physiological pH values in normal and tumor tissues. This was derived from the Boltzmann model fit (Fig. 2f) and a stability test showing a standard deviation ($\sigma$) of 0.0009 (Fig. 2g).
- Excellent reversibility was confirmed by a relative standard deviation (RSD) of 0.8% over five cycles (Fig. 2h), indicating consistent performance.
- Minimal leaching of HPTS-IP (1.6% decrease in fluorescence intensity over 72 hours, Fig. 2i) and excellent selectivity against various interfering substances (Fig. 2j) further validated its reliability in complex biological environments.
- Critically, negligible temperature crosstalk was observed in the pH range of 4.0 to 7.0, and for pH > 7.0, the probe's integrated temperature monitoring capability provides built-in compensation, ensuring accurate pH sensing (Fig. 2k).
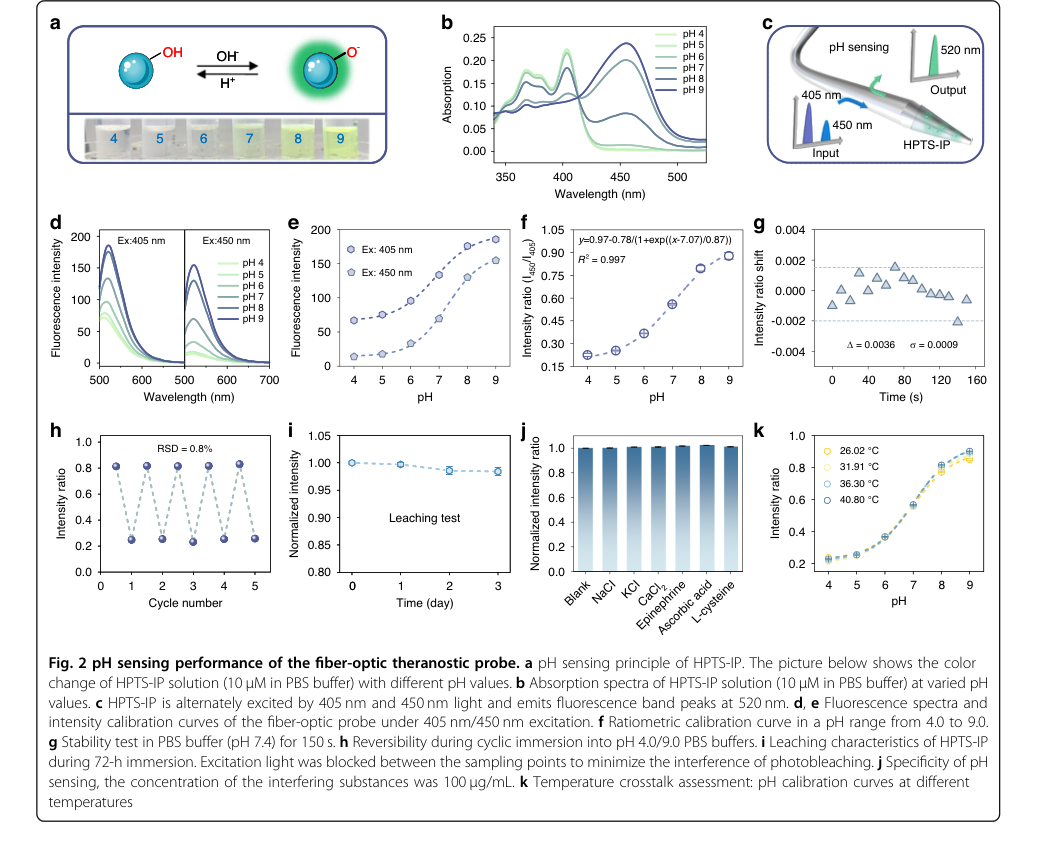 Figure 2. pH sensing performance of the fiber-optic theranostic probe. a pH sensing principle of HPTS-IP. The picture below shows the color change of HPTS-IP solution (10 μM in PBS buffer) with different pH values. b Absorption spectra of HPTS-IP solution (10 μM in PBS buffer) at varied pH values. c HPTS-IP is alternately excited by 405 nm and 450 nm light and emits fluorescence band peaks at 520 nm. d, e Fluorescence spectra and intensity calibration curves of the fiber-optic probe under 405 nm/450 nm excitation. f Ratiometric calibration curve in a pH range from 4.0 to 9.0. g Stability test in PBS buffer (pH 7.4) for 150 s. h Reversibility during cyclic immersion into pH 4.0/9.0 PBS buffers. i Leaching characteristics of HPTS-IP during 72-h immersion. Excitation light was blocked between the sampling points to minimize the interference of photobleaching. j Specificity of pH sensing, the concentration of the interfering substances was 100 μg/mL. k Temperature crosstalk assessment: pH calibration curves at different temperatures
Figure 2. pH sensing performance of the fiber-optic theranostic probe. a pH sensing principle of HPTS-IP. The picture below shows the color change of HPTS-IP solution (10 μM in PBS buffer) with different pH values. b Absorption spectra of HPTS-IP solution (10 μM in PBS buffer) at varied pH values. c HPTS-IP is alternately excited by 405 nm and 450 nm light and emits fluorescence band peaks at 520 nm. d, e Fluorescence spectra and intensity calibration curves of the fiber-optic probe under 405 nm/450 nm excitation. f Ratiometric calibration curve in a pH range from 4.0 to 9.0. g Stability test in PBS buffer (pH 7.4) for 150 s. h Reversibility during cyclic immersion into pH 4.0/9.0 PBS buffers. i Leaching characteristics of HPTS-IP during 72-h immersion. Excitation light was blocked between the sampling points to minimize the interference of photobleaching. j Specificity of pH sensing, the concentration of the interfering substances was 100 μg/mL. k Temperature crosstalk assessment: pH calibration curves at different temperatures
Temperature sensing also exhibited robust performance:
- The ratiometric approach, leveraging thermally enhanced energy transfer from Tb³⁺ to Eu³⁺ (Fig. 3a, c), allowed for precise monitoring.
- A resolution of 0.3 °C was achieved, satisfying in vivo monitoring requirements, with a linear relationship observed in the physiologically relevant range (34.5-39 °C) (Fig. 3i).
- High reversibility (RSD of 1.7% over 10 cycles, Fig. 3k) and minimal LnMOF leaching (3.5% decrease over 72 hours, Fig. 3l) were demonstrated.
- The evidence also proved its pH independence (Fig. 3m) and excellent specificity (Fig. 3n), ensuring accurate temperature readings regardless of pH fluctuations or other biological interferents.
The photothermal conversion capability of the probe was definitively established:
- Under only 267 mW excitation, the probe heated itself to 102.9 °C in vitro (Fig. 4b, c), confirming efficient photothermal conversion. This is a significant victory over conventional nanomaterial-based systems, which typically require pump powers greater than 1 W, making this approach more cost-effective and reducing potential side effects.
- Excellent photothermal stability was shown by stable temperature elevation over five heating/cooling cycles (Fig. 4d), and minimal ICG leaching (5.9% decrease over 72 hours, Fig. 4g, h) ensured long-term performance.
- In vivo experiments confirmed progressive temperature increase in tumor regions with rising pump power (Fig. 4e, f).
The in vivo validation provided the most compelling evidence for the probe's real-world utility:
- Tumor edge identification was unequivocally proven by a statistically significant difference in measured pH values between healthy and tumor tissues (p < 0.0001), and between tumor surface and normal tissue (p < 0.01) (Fig. 5a, b). This is definitive evidence of its diagnostic capability.
- Precise thermal dose control during PTT was demonstrated by actively maintaining the fiber temperature at 65 °C, which ensured the tumor edge reached an effective therapeutic temperature of 45 °C (Fig. 5c).
- Rapid efficacy assessment was validated by a significant pH elevation in the TME post-treatment (0.12-0.22 units increase within 30 minutes, and p < 0.01 at 24 hours) (Fig. 5d, e, f), serving as a reliable biomarker for therapeutic response.
- The ultimate proof of antitumor efficacy came from the tumor volume monitoring: 3 out of 4 treated mice showed complete tumor regression, with significant growth suppression in the remaining mouse, while the "victim" control group exhibited progressive tumor growth (Fig. 5g, h, i, j).
- Excellent biocompatibility and biosafety were confirmed by stable body weights in all mice (Fig. 5k), no significant apoptotic/necrotic areas or inflammatory cell infiltration in H&E stained organs (Fig. S10), and high cell viability in in vitro leachate tests (Fig. S9, Fig. 6c).
- Finally, multimodal anti-tumor effects were substantiated by histological analysis (Fig. 6a), revealing multifocal necrosis and hemorrhagic regions in treated tumors. Immunohistochemistry showed upregulated Caspase-3 (apoptosis biomarker), downregulated Ki67 (proliferation biomarker), and downregulated HIF-1α, indicating ameliorated tumor hypoxia, which was further corroborated by MRI (Fig. 6b, Fig. S8). This comprehensive evidence undeniably proves that the probe's core mechanism actually worked in reality, leading to effective tumor ablation and TME remodeling.
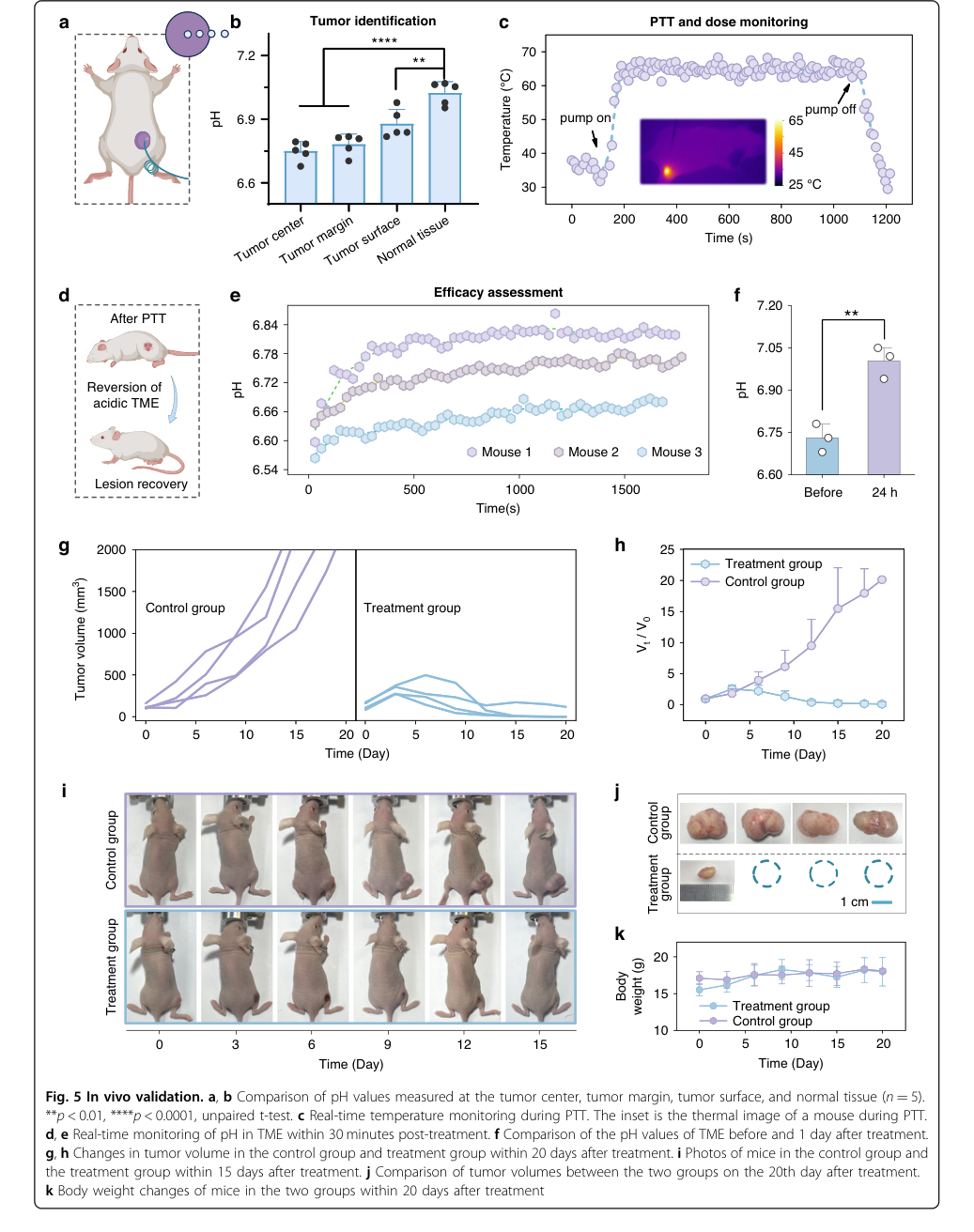 Figure 5. In vivo validation. a, b Comparison of pH values measured at the tumor center, tumor margin, tumor surface, and normal tissue (n = 5). **p < 0.01, ****p < 0.0001, unpaired t-test. c Real-time temperature monitoring during PTT. The inset is the thermal image of a mouse during PTT. d, e Real-time monitoring of pH in TME within 30 minutes post-treatment. f Comparison of the pH values of TME before and 1 day after treatment. g, h Changes in tumor volume in the control group and treatment group within 20 days after treatment. i Photos of mice in the control group and the treatment group within 15 days after treatment. j Comparison of tumor volumes between the two groups on the 20th day after treatment. k Body weight changes of mice in the two groups within 20 days after treatment
Figure 5. In vivo validation. a, b Comparison of pH values measured at the tumor center, tumor margin, tumor surface, and normal tissue (n = 5). **p < 0.01, ****p < 0.0001, unpaired t-test. c Real-time temperature monitoring during PTT. The inset is the thermal image of a mouse during PTT. d, e Real-time monitoring of pH in TME within 30 minutes post-treatment. f Comparison of the pH values of TME before and 1 day after treatment. g, h Changes in tumor volume in the control group and treatment group within 20 days after treatment. i Photos of mice in the control group and the treatment group within 15 days after treatment. j Comparison of tumor volumes between the two groups on the 20th day after treatment. k Body weight changes of mice in the two groups within 20 days after treatment
Limitations & Future Directions
While the presented fiber-optic theranostic probe demonstrates remarkable capabilities, it's important to acknowledge certain limitations and consider avenues for future development.
One inherent limitation, though mitigated by the fiber-optic design, is the light penetration depth. The evanescent field, while enabling localized sensing, has a shallow penetration depth (Fig. S5), meaning the probe primarily senses and treats the immediate vicinity of its surface. While this is suitable for precise, localized interventions, broader, volumetric mapping of the TME might require multiple probes or more advanced optical strategies. The response times for pH sensing (up to 12.8 s for decrease) and temperature sensing (up to 2.0 s for increase) are good for many applications but could be a limitation for capturing extremely rapid, millisecond-scale physiological dynamics.
Another practical consideration is leaching. Although the paper reports minimal leaching for HPTS-IP (1.6%), LnMOF (3.5%), and ICG (5.9%) over 72 hours, this is not zero. For long-term implantable devices, even minimal chronic leaching could accumulate or trigger immune responses over weeks or months, potentially affecting both probe performance and patient safety. The mention of blocking excitation light between sampling points to minimize photobleaching (Fig. 2i, 3l captions) suggests that while ratiometric detection helps, photobleaching remains a factor that could impact very long-duration continuous monitoring. Furthermore, while temperature crosstalk for pH sensing was compensated for pH > 7.0, it wasn't entirely negligible across the entire pH range, indicating a subtle interplay that might require more sophisticated compensation algorithms in the future. The choice of a single coating layer, while optimizing for both SNR and response time, implies a trade-off; thicker films could enhance signal but at the cost of slower response.
Looking forward, the paper itself proposes several exciting directions. The integration strategy can be translated to flexible polymer and hydrogel optical fibers to enhance mechanical compliance and biocompatibility, paving the way for long-term implantable devices. Leveraging spectral resources and employing multipeak fitting algorithms could further enhance functional capacity by addressing potential emission band overlap as more functions are integrated. The authors also suggest integrating the system with MRI thermometry for more accurate temperature monitoring of the tumor edge, implying that current thermal imaging might have limitations in spatial resolution or depth without this complement.
Beyond the paper's explicit suggestions, several discussion topics emerge for further development and evolution:
-
Advanced Multi-parameter Integration and AI-driven Feedback: How many more TME parameters (e.g., oxygen saturation, glucose levels, specific enzyme activities, drug concentrations) can be simultaneously integrated into a single fiber using WDM or other multiplexing techniques without compromising signal integrity or increasing device complexity? Can real-time data from such a probe be fed into an artificial intelligence (AI) or machine learning (ML) model to create truly adaptive, personalized treatment protocols that dynamically adjust laser power, duration, or even drug delivery based on the evolving TME? This could move beyond simple feedback loops to predictive, optimized therapy.
-
Long-term Biostability and Immunomodulation: While short-term biocompatibility is proven, what are the long-term implications of chronic implantation? How do biofouling, encapsulation by host tissue, or chronic inflammatory responses affect the probe's sensing accuracy and therapeutic efficacy over months or years? Can the sol-gel matrix be further engineered with immunomodulatory properties to actively promote integration and minimize adverse reactions, perhaps by releasing anti-inflammatory agents or growth factors?
-
Scalability, Miniaturization, and Clinical Workflow Integration: The current probe has a diameter of 440 µm, with a 100 µm tip. Can this be further miniaturized for even less invasive procedures, or can arrays of such probes be developed for larger or multifocal tumors? What are the manufacturing challenges and costs associated with scaling up production for widespread clinical use? How would such a sophisticated device integrate into existing clinical workflows, considering the need for specialized training, equipment, and data interpretation?
-
Non-invasive Guidance and Complementary Therapies: Could the principles of this closed-loop theranostic system be adapted to guide or enhance non-invasive therapies? For instance, could a less invasive version of the probe provide real-time TME feedback to optimize external radiation therapy, focused ultrasound, or systemic chemotherapy, rather than solely performing PTT? This would broaden its applicability beyond direct tumor ablation.
-
Ethical Considerations and Patient Experience: As these devices become more sophisticated and implantable, what are the ethical considerations regarding data privacy, patient autonomy, and the potential for over-treatment or unintended side effects from continuous, adaptive therapy? How can the patient experience be optimized, minimizing discomfort and psychological burden associated with implanted devices and frequent monitoring?
These discussions highlight the vast potential of this technology while underscoring the complex, multidisciplinary challenges that must be addressed for its full realization and widespread impact in oncology.
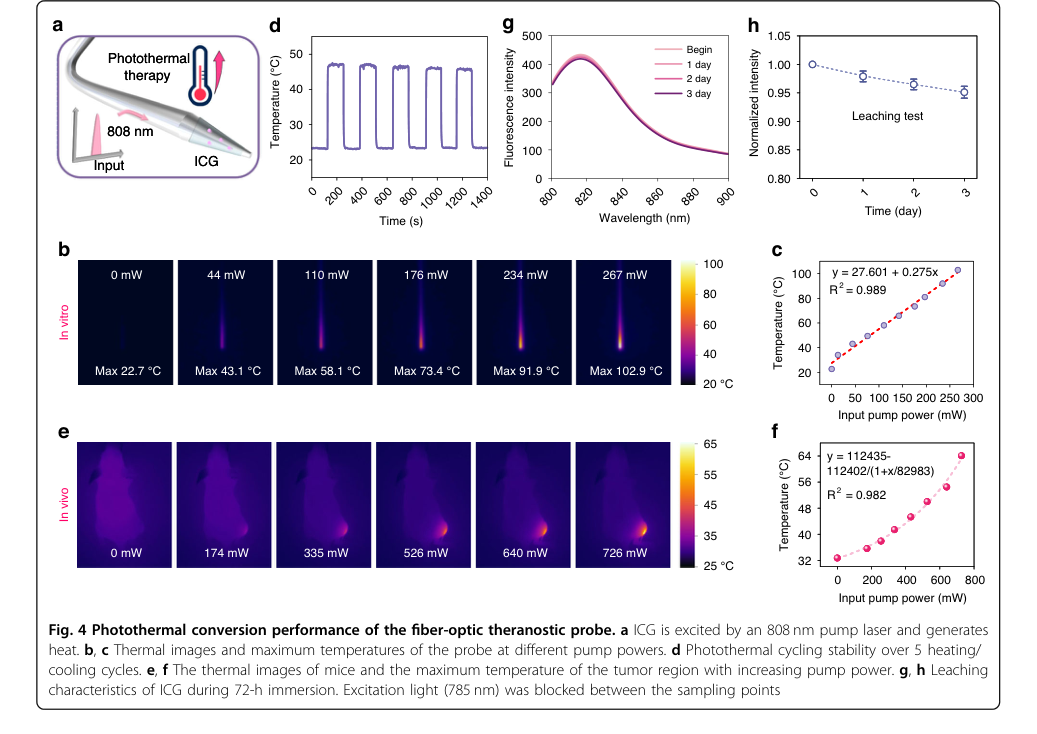 Figure 4. Photothermal conversion performance of the fiber-optic theranostic probe. a ICG is excited by an 808 nm pump laser and generates heat. b, c Thermal images and maximum temperatures of the probe at different pump powers. d Photothermal cycling stability over 5 heating/ cooling cycles. e, f The thermal images of mice and the maximum temperature of the tumor region with increasing pump power. g, h Leaching characteristics of ICG during 72-h immersion. Excitation light (785 nm) was blocked between the sampling points
Figure 4. Photothermal conversion performance of the fiber-optic theranostic probe. a ICG is excited by an 808 nm pump laser and generates heat. b, c Thermal images and maximum temperatures of the probe at different pump powers. d Photothermal cycling stability over 5 heating/ cooling cycles. e, f The thermal images of mice and the maximum temperature of the tumor region with increasing pump power. g, h Leaching characteristics of ICG during 72-h immersion. Excitation light (785 nm) was blocked between the sampling points
